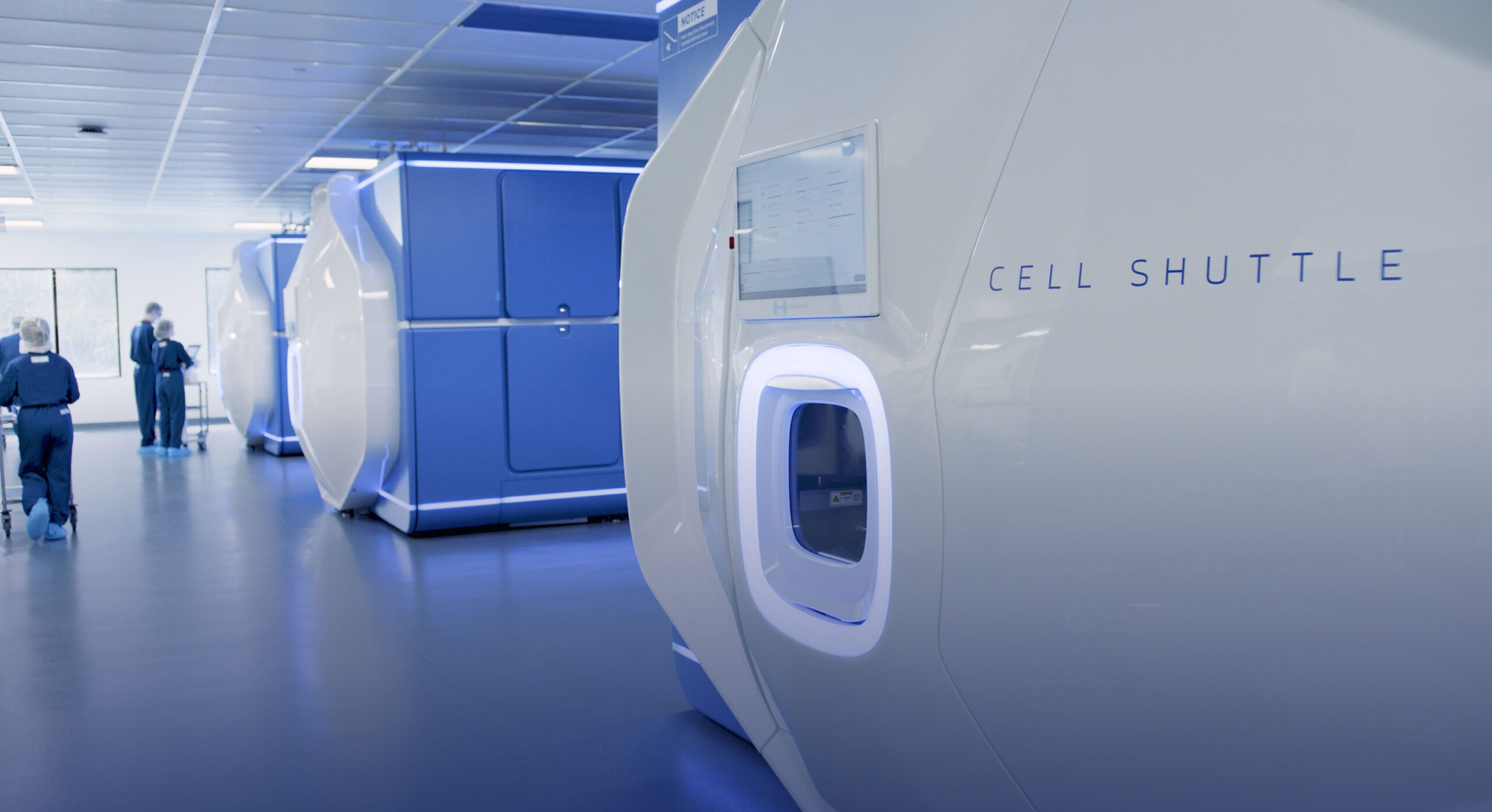
Cellares’ Mission: Meet Total Patient Demand
by Delivering Cell Therapy Manufacturing Excellence At Scale Across a Global Network of IDMO Smart Factories

-
Smart Factory
South San Francisco, CA - Preclinical & Clinical Services / Technology Development
- 57,000 Sq Ft

-
Smart Factory
South San Francisco, CA - Preclinical & Clinical Services / Technology Development
- 57,000 Sq Ft

-
Smart Factory
Bridgewater, NJ - Preclinical, Clinical & Commercial Services
- 118,000 Sq Ft

-
Smart Factory
Bridgewater, NJ - Preclinical, Clinical & Commercial Services
- 118,000 Sq Ft

-
Smart Factory - Europe
Leiden, Netherlands - Preclinical, Clinical & Commercial Services
- 100,000 Sq Ft

-
Smart Factory - Europe
Leiden, Netherlands - Preclinical, Clinical & Commercial Services
- 100,000 Sq Ft

-
Smart Factory - Asia
Kashiwa, Japan - Preclinical, Clinical & Commercial Services
- 150,000 Sq Ft

-
Smart Factory - Asia
Kashiwa, Japan - Preclinical, Clinical & Commercial Services
- 150,000 Sq Ft
- Advanced Technology
-

- IDMO Services
Operational Excellence Built Into Our Integrated Development and Manufacturing Organization (IDMO) Infrastructure

IDMO Capabilities
-

Process Translation & Automation
Partner with our team to translate your process onto the AMT-designated Cell Shuttle, achieving 100% process automation while preserving your existing drug product quality attributes and performance.
-

Analytical Bridging & Automation
Partner with our team to automate your analytical methods using the cGMP-compliant Cell Q, accelerating method transfer, qualification, and validation, while ensuring data integrity and continuous compliance.
-

Global Quality & Regulatory Consultation
Leverage the AMT award-winning team to accelerate timelines from pre-IND to BLA, with full CMC alignment and harmonization across major Health Authorities.
Cellares’ IDMO Advantage
Designed to Outperform Any Manual In-House or CDMO Operation on Scalability, Cost of Manufacturing, Quality, and Speed to Market
-

Scalability
Automated Smart Factories have 10x the productivity of manual facilities which enables rapid capacity expansion without the constraints. This unlocks revenue and ensures supply can meet global demand.
-

Cost Savings
Highly automated operations reduce manufacturing costs and improve margins. Cellares delivers one of the lowest cost structures in the industry.
-

Quality
Automation minimizes human error and contamination risk, enabling consistent execution across batches and sites. This increases reliability, product quality, and partner’s market share.
-

Speed to Market
Digital process and method transfer and a global Smart Factory network enable rapid expansion into new regions. This can accelerate market entry by 3 to 4 years and maximize on-patent revenue.
-

Predictable Cost Structure
Automation enables transparent per-batch pricing with no hidden fees. This provides long-term cost visibility and predictability not possible with traditional CDMOs.
-

Peak Demand Coverage
Cellares can absorb fluctuations in commercial demand, enabling partners to maintain efficient, predictable utilization of internal capacity without over-investing in infrastructure and headcount to meet peak requirements.
Recent News
-
 April 28, 2026
April 28, 2026Cellares and Cabaletta Bio Sign 10-Year Commercial Supply Agreement to Scale Rese-cel
-
 April 14, 2026
April 14, 2026First Patients Dosed with Cabaletta Bio’s Rese-cel Manufactured on Cellares’ Automated Cell Shuttle™ Platform
-
 May 11, 2026
May 11, 2026Cellares and ProTgen Partner to Automate Manufacturing for ProT-096, a Novel Progenitor T Cell Therapy Designed to Restore Immune Competence