
The Cellares IDMO overcomes the limitations of manual CDMOs
Traditional CDMOs rely on manual processes that limit scalability and increase cost and process failure rates
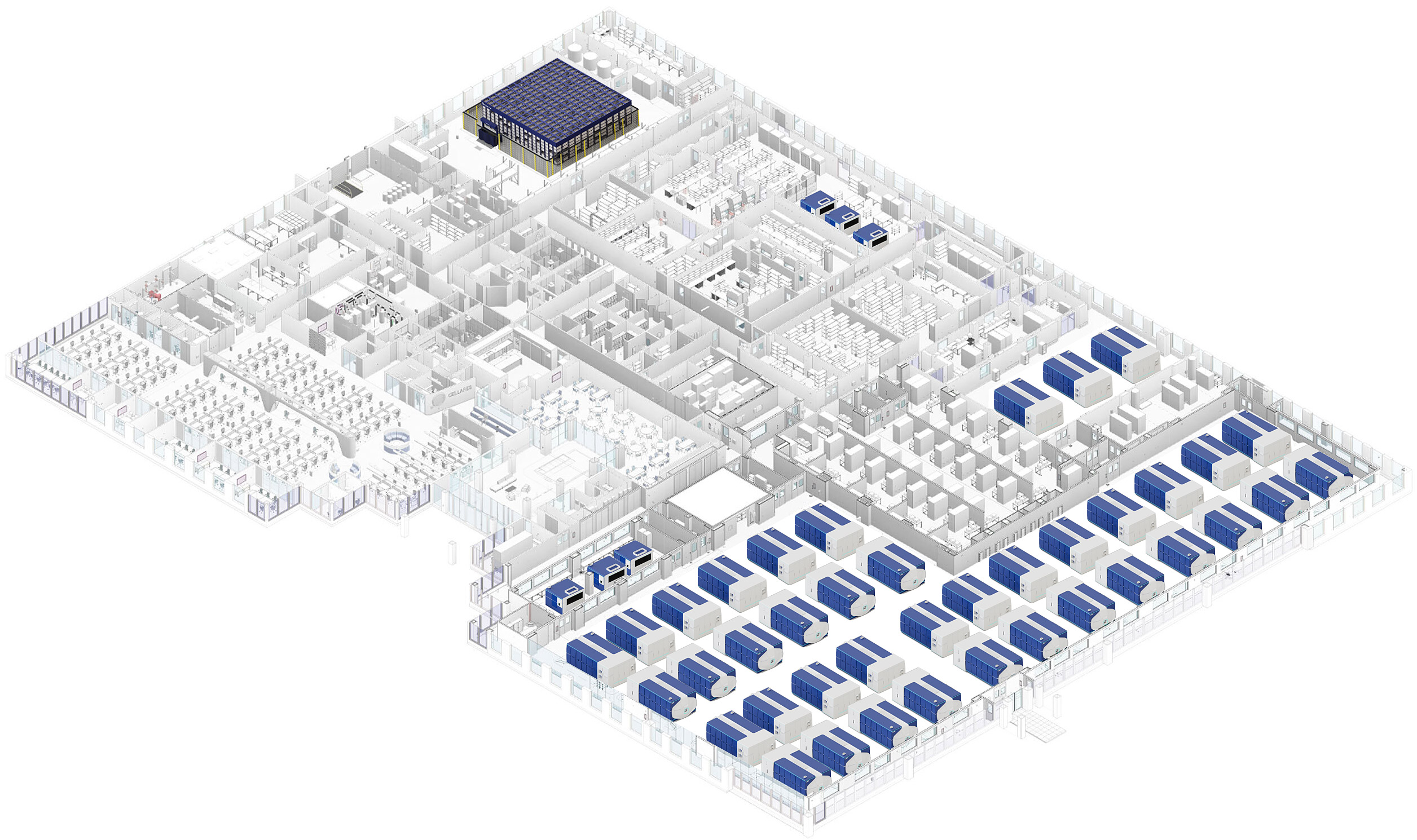
Cellares pioneered the Integrated Development and Manufacturing Organization (IDMO), combining digitally connected and automated manufacturing platforms with global Smart Factory infrastructure
Cellares pioneered the Integrated Development and Manufacturing Organization (IDMO), combining digitally connected and automated manufacturing platforms with global Smart Factory infrastructure
-
215KGlobal Batch CapacityCellares' global network of Smart Factories is purpose-built to meet total patient demand at scale across clinical and commercial programs worldwide.
-
90%Reduction in Facility SizeThe Cell Shuttle is functionally equivalent to ~100 benchtop instruments, but only takes up 10% of the floor space that 100 benchtop instruments would take up.
-
90%Reduction in LaborEnd-to-end automation across process and analytics reduces direct labor requirements by up to 90%, decreasing reliance on operators, lowering training costs, and eliminating the risk of knowledge loss due to staff turnover.
IDMO Drug Product Journey
From patient material receipt to Certificate of Analysis generation, every step is barcode-tracked and electronically recorded, ensuring complete chain of identity, data integrity, and a fully auditable manufacturing record
IDMO Smart Factory
The Cellares Smart Factory integrates best-in-class automation across supply chain, inventory, media/reagent filling, fill-finish, and cryostorage, all tied into a unified digital backbone spanning ERP/LIMS, eBR, and COI/COC, delivering low-touch execution, real-time visibility, and a continuous state of compliance where every action is traceable.
Automated Cryostorage
- From drug product formulation to QC vial storage, automated cryostorage ensures safe, traceable, and temperature-controlled handling of every patient batch, maintaining chain of identity from manufacturing through release.
- Flexible cryostorage configurations support both clinical and commercial programs, with barcode-enabled tracking and automated retrieval ensuring accurate, error-proof handling of temperature-sensitive materials at every stage.
Automated Cryostorage
- From drug product formulation to QC vial storage, automated cryostorage ensures safe, traceable, and temperature-controlled handling of every patient batch, maintaining chain of identity from manufacturing through release.
- Flexible cryostorage configurations support both clinical and commercial programs, with barcode-enabled tracking and automated retrieval ensuring accurate, error-proof handling of temperature-sensitive materials at every stage.
Automated and High-Throughput Cell Therapy Quality Control
- The Cell Q platform automates end-to-end quality control, from barcode-tracked sample preparation and assay execution to data capture and Certificate of Analysis generation, supporting up to 6,000 batches per year with consistent, audit-ready results.
- Integrating best-in-class analytical instruments with automated liquid handling, Cell Q standardizes cell counting, flow cytometry, and potency and safety testing, reducing operator-dependent variability and accelerating batch release.
Automated and High-Throughput Cell Therapy Quality Control
- The Cell Q platform automates end-to-end quality control, from barcode-tracked sample preparation and assay execution to data capture and Certificate of Analysis generation, supporting up to 6,000 batches per year with consistent, audit-ready results.
- Integrating best-in-class analytical instruments with automated liquid handling, Cell Q standardizes cell counting, flow cytometry, and potency and safety testing, reducing operator-dependent variability and accelerating batch release.
Automated Raw Material Management
- Cellares’ automated warehouse and integrated ERP system reduces manual touchpoints, provides real-time inventory visibility, and ensures end-to-end traceability from raw material receipt through manufacturing.
- Barcode-based tracking of every material and reagent enables predictable consumption, reduces waste, and maintains a continuous, audit-ready inventory record across the entire manufacturing operation.
Automated Raw Material Management
- Cellares’ automated warehouse and integrated ERP system reduces manual touchpoints, provides real-time inventory visibility, and ensures end-to-end traceability from raw material receipt through manufacturing.
- Barcode-based tracking of every material and reagent enables predictable consumption, reduces waste, and maintains a continuous, audit-ready inventory record across the entire manufacturing operation.
Automated Bulk Reagent Filling
- Reagents are prepared under ISO 5 conditions, bulk-filled into barcoded reagent bottles, and delivered automatically to the corresponding patient batch via the Cell Shuttle's temperature-controlled Reagent Vault System.
- Closed, aseptic reagent preparation with software-controlled delivery ensures consistent, scalable throughput while reducing the risk of contamination.
Automated Bulk Reagent Filling
- Reagents are prepared under ISO 5 conditions, bulk-filled into barcoded reagent bottles, and delivered automatically to the corresponding patient batch via the Cell Shuttle's temperature-controlled Reagent Vault System.
- Closed, aseptic reagent preparation with software-controlled delivery ensures consistent, scalable throughput while reducing the risk of contamination.
Automated and High-Throughput Cell Therapy Manufacturing
- The FDA AMT-designated Cell Shuttle delivers industrial-scale, cGMP cell therapy manufacturing in a closed, automated ISO 8 environment, with single-use, barcoded consumables supporting up to 2,500 batches per system per year.
- All unit operations, from magnetic selection and transduction to expansion and harvest, are fully automated within a single-use consumable cartridge, eliminating manual interventions and reducing contamination risk.
Automated and High-Throughput Cell Therapy Manufacturing
- The FDA AMT-designated Cell Shuttle delivers industrial-scale, cGMP cell therapy manufacturing in a closed, automated ISO 8 environment, with single-use, barcoded consumables supporting up to 2,500 batches per system per year.
- All unit operations, from magnetic selection and transduction to expansion and harvest, are fully automated within a single-use consumable cartridge, eliminating manual interventions and reducing contamination risk.

Global Headquarters & Clinical Manufacturing Site
South San Francisco, CA
2K - 10K Doses / Year
2K - 10K Doses / Year
In cell therapy, process scaling is where clinical promise often collides with manufacturing reality: manual operations expose hidden variability during scale-up, while lengthy site-to-site transfers add to comparability risk.
At Cellares, we encode Quality by Design (QbD) directly into the Cell Shuttle so process knowledge is digitized and continuously refined, from translation and automation through optimization and characterization. This reduces reliance on “experienced operators,” ensures consistent performance, and generates reliable datasets for late-stage risk assessment and lifecycle management.
When it’s time to scale, our closed, automated systems standardize operations across facilities, delivering lower batch-to-batch variability and comparability by design. A platform-based transfer activates new sites in months, not years, avoiding the costly re-development cycles typical of conventional CDMO engagements.
At Cellares, we encode Quality by Design (QbD) directly into the Cell Shuttle so process knowledge is digitized and continuously refined, from translation and automation through optimization and characterization. This reduces reliance on “experienced operators,” ensures consistent performance, and generates reliable datasets for late-stage risk assessment and lifecycle management.
When it’s time to scale, our closed, automated systems standardize operations across facilities, delivering lower batch-to-batch variability and comparability by design. A platform-based transfer activates new sites in months, not years, avoiding the costly re-development cycles typical of conventional CDMO engagements.

Commercial Manufacturing - US
Bridgewater, NJ
20K - 100K Doses / Year
20K - 100K Doses / Year
Transitioning from reliable clinical supply to repeatable commercial output demands more than capacity. It requires an infrastructure built for consistency.
Cellares' flagship IDMO Smart Factory integrates best-in-class automation spans supply chain, inventory, media/reagent filling, fill-finish, and cryostorage, all tied into a unified digital backbone spanning ERP/LIMS, eBR, and COI/COC, delivering low-touch execution, real-time visibility, and a continuous state of compliance where materials move, data follows, and every action is traceable.
Strategically located near major academic medical centers, research institutions, and regulatory bodies, the facility supports efficient clinical and commercial supply while ensuring seamless process continuity across the Cellares global network.
When it's time to expand globally, the same proven blueprint repeats: standardized operations, digitized processes, and vendor-validated interfaces activate new sites in months, not years, delivering operational excellence from day one.
Cellares' flagship IDMO Smart Factory integrates best-in-class automation spans supply chain, inventory, media/reagent filling, fill-finish, and cryostorage, all tied into a unified digital backbone spanning ERP/LIMS, eBR, and COI/COC, delivering low-touch execution, real-time visibility, and a continuous state of compliance where materials move, data follows, and every action is traceable.
Strategically located near major academic medical centers, research institutions, and regulatory bodies, the facility supports efficient clinical and commercial supply while ensuring seamless process continuity across the Cellares global network.
When it's time to expand globally, the same proven blueprint repeats: standardized operations, digitized processes, and vendor-validated interfaces activate new sites in months, not years, delivering operational excellence from day one.

Clinical & Commercial Manufacturing - Europe
Leiden, Netherlands
6K - 30K Doses / Year
6K - 30K Doses / Year
Cellares’ European headquarters in Leiden expands the company’s global Smart Factory network with regional manufacturing capacity for European patients.
Located at Leiden Bio Science Park, the facility will provide biopharmaceutical partners with access to Cellares’ automated Cell Shuttle™ manufacturing platform and Cell Q™ quality control technologies, enabling scalable, standardized, and reliable production closer to patients.
Leiden offers a strong Life Sciences & Health ecosystem, bringing together leading biotech companies, research institutions, and manufacturing expertise in a highly connected environment.
This location supports consistent process execution across geographies while enabling efficient regional supply for clinical and commercial programs.
Located at Leiden Bio Science Park, the facility will provide biopharmaceutical partners with access to Cellares’ automated Cell Shuttle™ manufacturing platform and Cell Q™ quality control technologies, enabling scalable, standardized, and reliable production closer to patients.
Leiden offers a strong Life Sciences & Health ecosystem, bringing together leading biotech companies, research institutions, and manufacturing expertise in a highly connected environment.
This location supports consistent process execution across geographies while enabling efficient regional supply for clinical and commercial programs.

Clinical & Commercial Manufacturing - Japan
Kashiwa, Japan
15K - 75K Doses / Year
15K - 75K Doses / Year
Cellares' first Smart Factory in the Asia Pacific region deliver regional manufacturing capacity for Japanese and broader APAC patients.
Located within one of the world's leading life sciences ecosystems, the facility provides biopharmaceutical partners with access to the Cell Shuttle manufacturing platform and Cell Q quality control technologies, enabling locally compliant, scalable, and standardized cell therapy production.
Japan's robust regulatory framework and deep biotechnology expertise make it an ideal hub for regional clinical and commercial programs, supporting consistent process execution and efficient patient supply across Asia Pacific markets.
Located within one of the world's leading life sciences ecosystems, the facility provides biopharmaceutical partners with access to the Cell Shuttle manufacturing platform and Cell Q quality control technologies, enabling locally compliant, scalable, and standardized cell therapy production.
Japan's robust regulatory framework and deep biotechnology expertise make it an ideal hub for regional clinical and commercial programs, supporting consistent process execution and efficient patient supply across Asia Pacific markets.