Proven Performance
Cellares has successfully automated a wide range of cell therapy manufacturing processes while meeting each partner’s success criteria
-
10+PartnershipsSuccessfully automated processes from pharma, biotech, and academic translation centers from preclinical programs to commercially approved therapies.
-
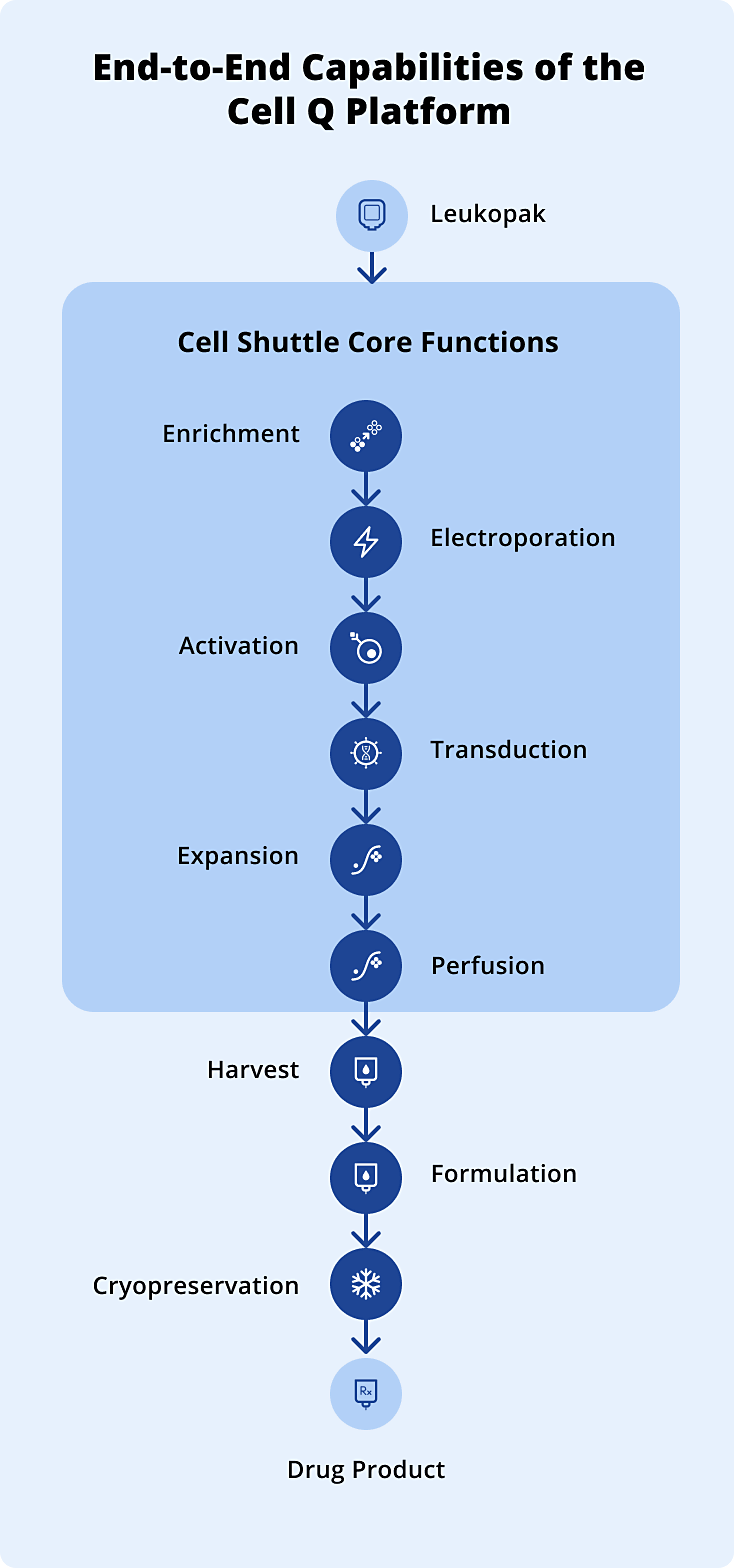
14+Unique ProcessesSuccessful automation of CAR-T, TCR-T, and HSC manufacturing processes using multiple gene delivery approaches, including viral vectors, electroporation, and lipid nanoparticles.
-
1000+Automated RunsReliable end-to-end execution of diverse processes using the same universal consumable cartridge design.
-
1400+Automated AssaysReliable end-to-end execution of client-specific drug product release assays using the high-throughput Cell Q platform.
Recent Partnerships
Clinical & Commercial Manufacturing
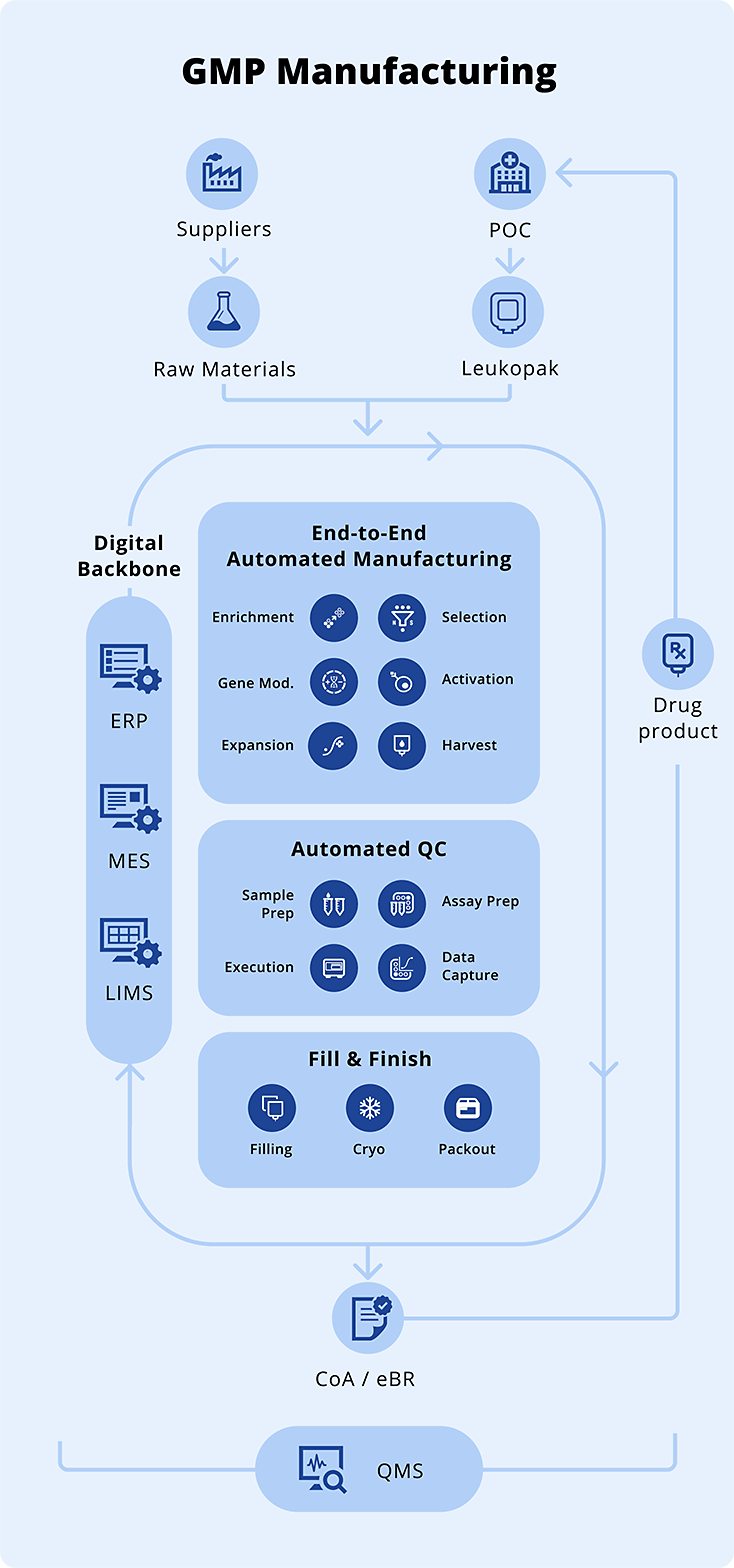
Operational Excellence Built Into Our Integrated Development and Manufacturing Organization (IDMO) Infrastructure
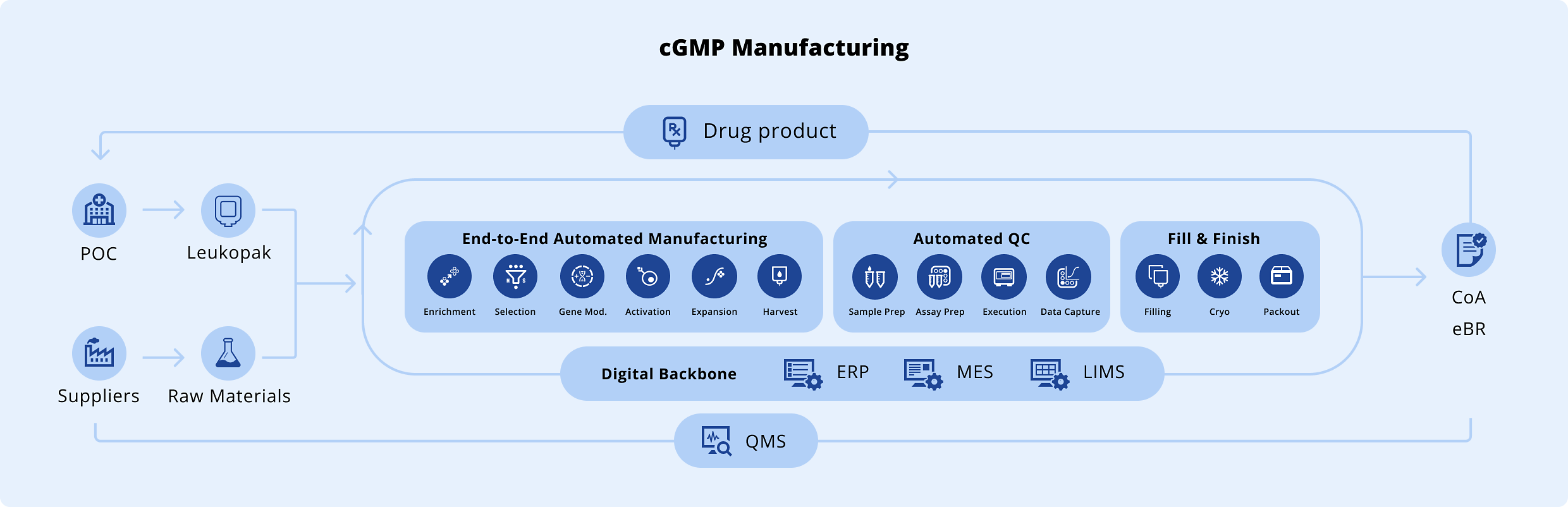
-
 Operational EfficiencyAutomation eliminates unnecessary manual touchpoints and enabling consistent, scalable performance across clinical and commercial manufacturing.
Operational EfficiencyAutomation eliminates unnecessary manual touchpoints and enabling consistent, scalable performance across clinical and commercial manufacturing. -
 ReliabilityContinuous process monitoring and automated controls deliver reliable, reproducible performance, ensuring every patient batch meets the highest quality standards.
ReliabilityContinuous process monitoring and automated controls deliver reliable, reproducible performance, ensuring every patient batch meets the highest quality standards. -
 Traceability & ComplianceEnd-to-end barcode tracking and automated data capture ensure a complete, auditable record for every patient batch and support regulatory compliance across sites.
Traceability & ComplianceEnd-to-end barcode tracking and automated data capture ensure a complete, auditable record for every patient batch and support regulatory compliance across sites.
"Having commercialized two CAR-T products, I know that operational excellence means reliable execution — every batch, every time. Our IDMO model brings together automated manufacturing, quality control, and digital infrastructure into one seamless operation, purpose-built for scale, consistency, and compliance from day one."
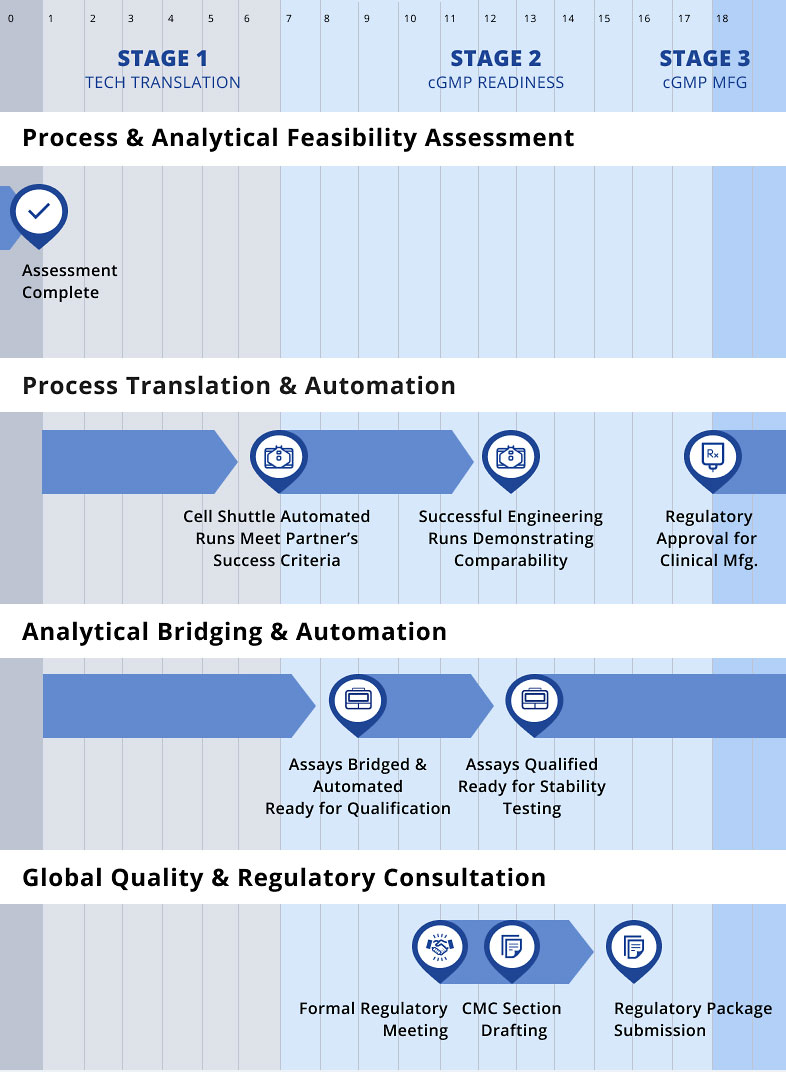
IDMO Journey
Cellares accelerates partner’s cell therapy programs from process translation through clinical and commercial manufacturing
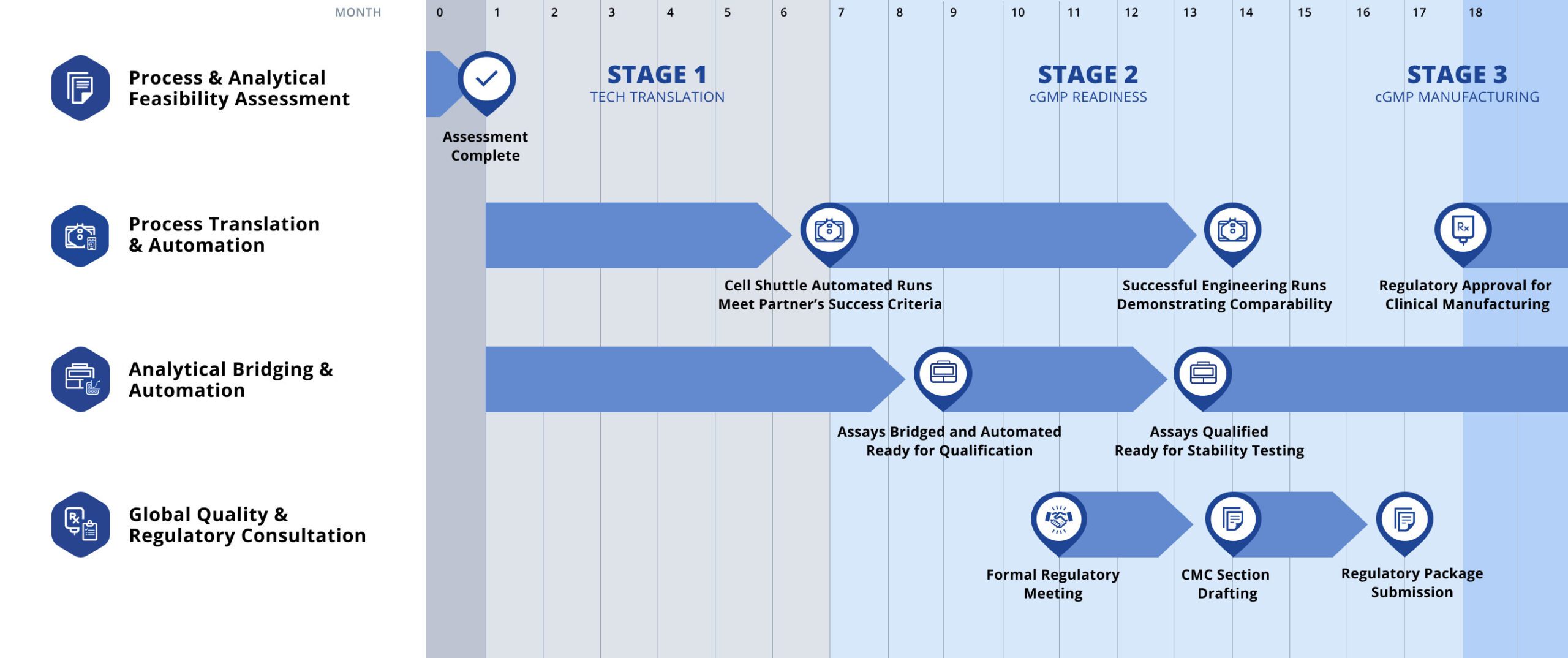
IDMO Capabilities
A new category of partnership combining automation technology, manufacturing capacity, and global infrastructure to expedite your path to commercially viable cell therapies
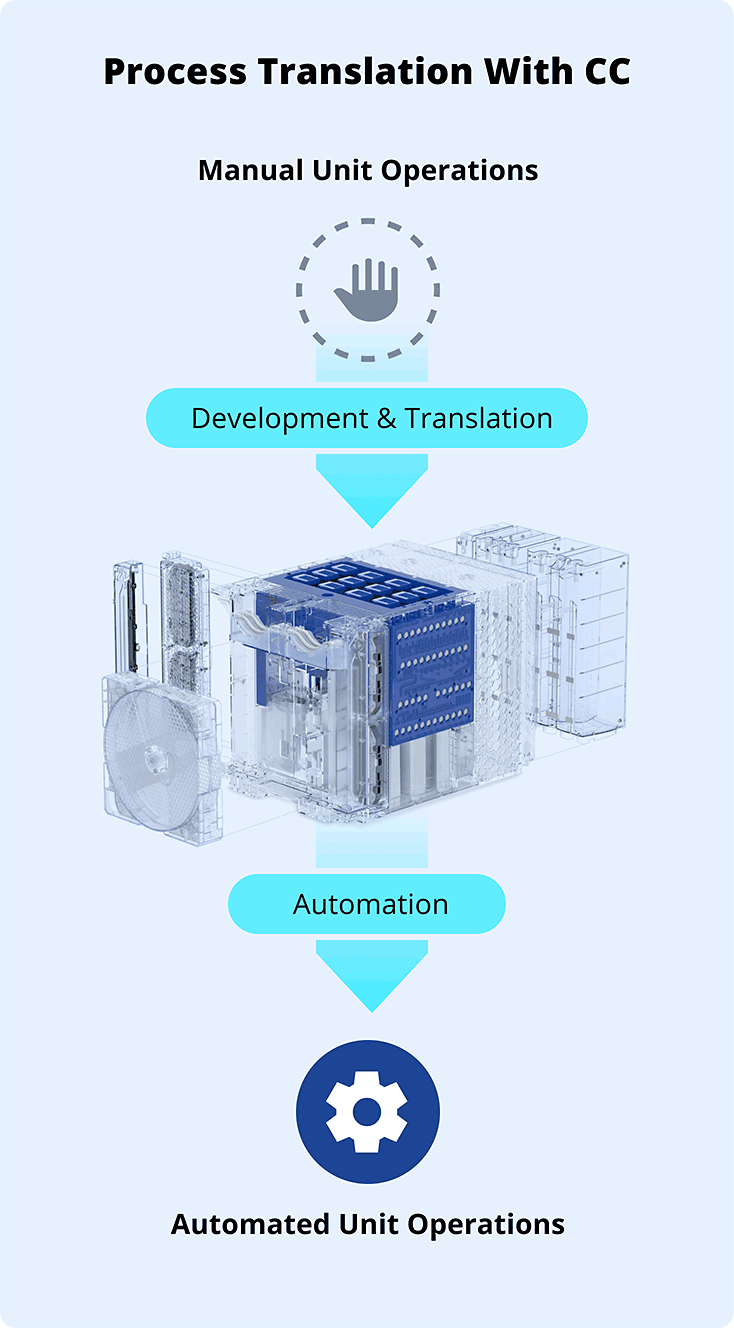
Process Translation & Automation
As your manufacturing partner, Cellares collaborates closely with partner’s team to translate and automate existing processes onto the Cell Shuttle platform, preserving critical quality attributes and ensuring analytical comparability.
A dedicated Alliance Manager is assigned to every partnership, ensuring transparent communication, milestone tracking, and proactive regulatory updates throughout your IDMO journey.
A dedicated Alliance Manager is assigned to every partnership, ensuring transparent communication, milestone tracking, and proactive regulatory updates throughout your IDMO journey.

-
 TransparencyAccess to all process parameters, such as electroporation settings. No royalty or licensing fees.
TransparencyAccess to all process parameters, such as electroporation settings. No royalty or licensing fees. -
 FlexibilityFine tune process parameters to achieve analytical comparability between manual and automated process.
FlexibilityFine tune process parameters to achieve analytical comparability between manual and automated process. -
 Quality by DesignProcess knowledge is encoded directly into the Cell Shuttle software, ensuring reliable design of experiment dataset.
Quality by DesignProcess knowledge is encoded directly into the Cell Shuttle software, ensuring reliable design of experiment dataset.
Analytical Bridging & Automation
As your manufacturing partner, Cellares collaborates closely with your scientific team to transfer and automate your existing manual analytical workflows onto the Cell Q platform — combining automated technologies with deep cell therapy expertise to ensure assay comparability and regulatory readiness.
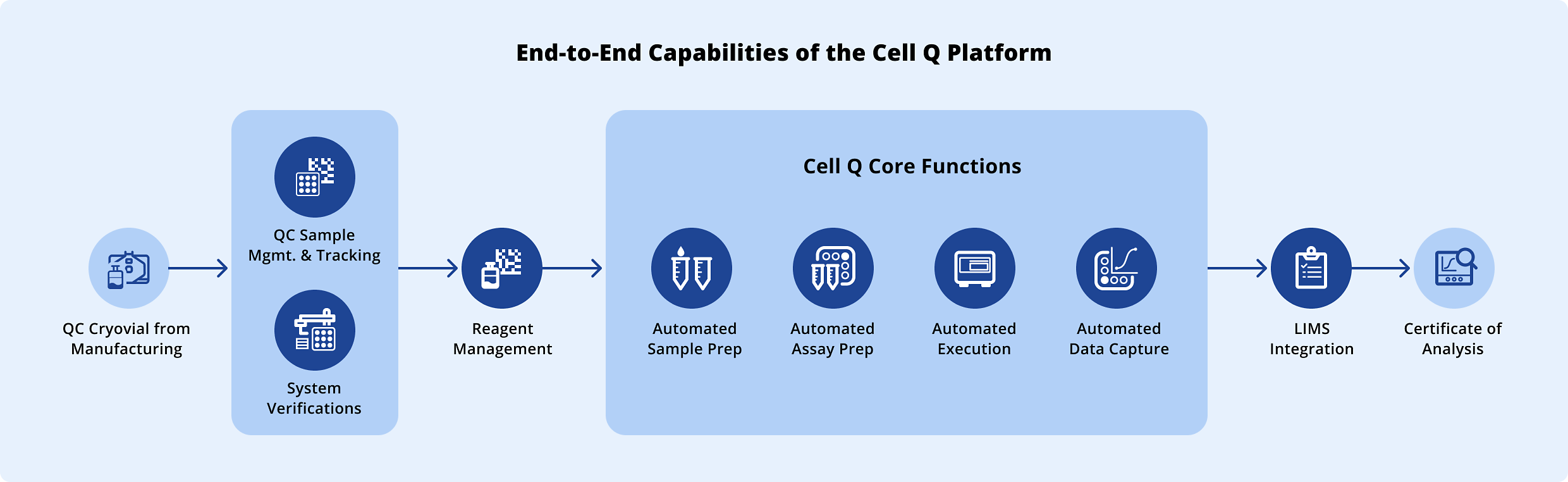
-
 TraceabilityData flows directly from instruments into secure data systems and is linked to the electronic batch record
TraceabilityData flows directly from instruments into secure data systems and is linked to the electronic batch record -
 ConsistencyAutomation enables consistent QC execution, streamlined batch record review, and inspection-ready documentation.
ConsistencyAutomation enables consistent QC execution, streamlined batch record review, and inspection-ready documentation. -
 ThroughputEach Cell Q can support 3000 to 6000 patient batch release per year, pending method complexity.
ThroughputEach Cell Q can support 3000 to 6000 patient batch release per year, pending method complexity.
We believe that Cellares’ automated manufacturing and quality control platforms have the potential to change the autologous cell therapy paradigm by offering the promise of unprecedented scale produced reliably with minimal capital expense.
Global Quality & Regulatory Consultation
Collaboratively develop and execute a global regulatory strategy aligned with your process and analytical program. Cellares’ FDA Advanced Manufacturing Technology (AMT) designation enables more frequent and prioritized interactions with the FDA. This supports early alignment, reduces regulatory risk, and streamlines the path to approval.

-
 Strategic PlanningBuild a cohesive CMC and regulatory strategy that leverages automation technologies, reducing rework and aligning with global expectations
Strategic PlanningBuild a cohesive CMC and regulatory strategy that leverages automation technologies, reducing rework and aligning with global expectations -
 Submission SupportSupport for authoring Briefing Books, Module 3, IR responses. Cellares will also issue LoAs to reference Master Files and AMT Designation, as applicable.
Submission SupportSupport for authoring Briefing Books, Module 3, IR responses. Cellares will also issue LoAs to reference Master Files and AMT Designation, as applicable. -
 Global Expansion ReadinessGlobal regulatory risk register and inspection readiness, anchored in GxP eQMS data integrity
Global Expansion ReadinessGlobal regulatory risk register and inspection readiness, anchored in GxP eQMS data integrity